■高せん断ミキサーによる湿式造粒を監視する為のNIRの適用
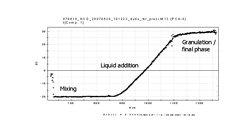
This typical pharmaceutical production process is a fast process, which is known for producing very dense granules resulting in a slow release of the API in the body. The process consists of
three phases a) dry mixing, b) liquid addition and c) massing or granulation.
The granulation process is responsible for major tablet properties as well as the processibility of the powders. The process is performed to achieve a granulation that provides the optimum tablet
hardness, dissolution/disintegration, friability, flowability when the granules are dry, compression quality, and content uniformity.
|
|
Why use NIR technology to monitor HSWG?
NIR spectroscopy is the only analyzer technology which provides deep insight about the physical and chemical properties during the process at very high measurement speed. It can detect changes in the particle size of excipients or API during the mixing phase and detect the end point based on the homogeneity in this phase, variation in particle growth throughout the process. Free and bound moisture can be monitored, since they have different effects on the NIR spectra. The monitoring of the dissolution of sugars during the granulation could be followed as well.

|
|
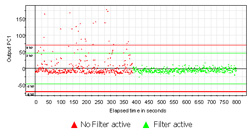
Why Diode array technology
Diode array spectrometers are much faster than other available technologies. In our optimized SentroPAT diode array systems a typical integration time is only approx. 10 ms giving an overall measurement interval of 1 sec or less. But even further, a diode array NIR detects all wavelengths of the spectrum simultaneously. This is totally different to all other NIR technologies including FT-NIR. When sample presentation is not constant during the process, (typical in the case of smaller granulators,) every single spectrum looks smooth. Only the baseline might be at different heights. Fast variations in a scanning spectrometer would make the spectrum unusable or show only noise. Similar effects reduce the data quality in FT-NIR systems.
|
|
Use cases of NIR in high shear wet granulation
Formulation development
If NIR is consequently used during formulation development it can generate much data, which can be used to compare the process behaviour and results for the complete development process. This
gained data can be used to create models for end point detection or to optimize the process parameters.
Process transfer
NIR can be used to support the transfer of a production process between different equipment and/or sites. Using the process data from the system in early development can be compared to the data
generated on systems in later stages. Possible variations can be spectroscopically analyzed more easily to find the root causes for different process results.
Process scale up
When NIR data from small scale are available at the beginning of a process scale up, it can be used to compare with the data from the larger scale for an immediate check on the process. Combined
process and NIR knowledge can efficiently support and speed up the overall process of scale up.
Production process
It is the goal to achieve process repeatability and consistent process results with reduced variability of the granulated mixture. The final goal is the development of an active process control
to ensure the output quality of the granulation in a variation that is as small as possible.
